 The electron configuration of Cobalt will end in d block as Cobalt is an inner transition metal. The electron configuration for Cobalt at ground state would simply be Co: Ar 4s23d7. 
The first electron has the same four quantum numbers as the hydrogen atom electron ( n 1, l 0, ml 0, ms +12 m s + 1 2 ). The helium atom contains two protons and two electrons. 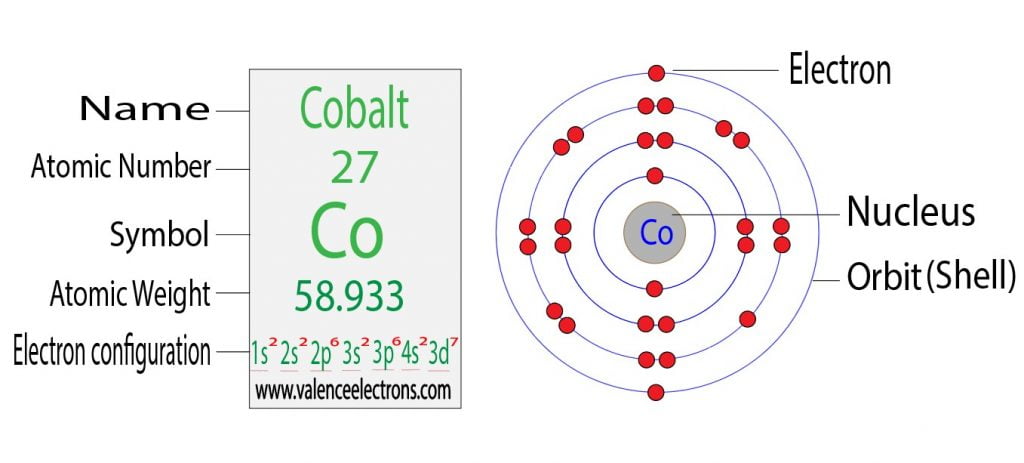
mar 2023 Electron Configuration Chart of All Elements (Full Chart) Electron. A: The electron configuration of a transition metal cation is written by removing the electrons first. The oxidation state of the element changes depending on the bond formation. The s,p,d,f configuration for cobalt (Co) is 1s22s22p63s23p64s23d7, determined by the position of the element on the periodic table. In a D4h site with a strong elongation the 3A2g term of cobalt (+III) can be stabilized with an electronic configuration intermediate between low spin. The electron configuration and the orbital diagram are: Following hydrogen is the noble gas helium, which has an atomic number of 2. Cobalt 2+ configuration Why does Co2+ have 7 electrons in the 3d orbital. Q: Explain how to write an electron configuration for a transition metal cation. Copper atoms exhibit +1 and +2 oxidation states. However, the electron configuration referred in this study was one that derived from the later atomic model the quantum mechanics atomic model. The concept of electron configuration has been introduced since the discovery of Bohr atomic model. The electron configuration of copper ions (Cu 2+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 9. Electron configuration describes the distribution of electrons within an atom. All elements can be represented in this fashion. Atomic number, atomic weight and charge of copper ion. c24 N69-32515 COEFFICIENTS Electronic configuration related to radiation. For example, the electron configuration of lithium, 1s2s, tells us that lithium has two electrons in the 1s subshell and one electron in the 2s subshell. The electron configuration of Cobalt is: 1s2 2s2 2p6 3s2 3p6 3d7 4s2. network configuration, and amplifiers in directly coupled, wideband. 
And if I remember correctly this has nothing to do with stabilization in ligand fields.\right] 5s^2\). Electron configurations describe where electrons are located around the nucleus of an atom. Well, you can find some kind of explanation why the electronic configuration of $\ce$ atom, they are lost from the 4$s$ orbital first because it is actually higher in energy when both $3d$ and $4s$ are filled with electrons. Nickel has the atomic number 28 so it has the electron configuration 1s22s22p63s23p64s23d8. Ground state electron configurations are the foundation for understanding molecular bonding, properties, and structures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
